Michele Carbonelli Department of Biomedical and Neuromotor Sciences (DIBINEM), University of Bologna, Bologna, ItalyStudio Oculistico d’Azeglio, Bologna, Italy
A 16-year-old boy, belonging to an Italian family affected by LHON, complained of subacute, painless visual loss in his right eye (OD). Three days after symptoms onset he presented with visual acuity (VA) OD of 0.2 and the ophthalmoscopy revealing small retinal vascular telangiectasias as well as pseudo-edema of the optic nerve head. A month later he complained of subacute visual loss also in his left eye (OS) associated with a small ceco-central scotoma on visual fields (VF) (Figure 1). Genetic evaluation was positive for the 11778/ND4 mtDNA pathogenic variant in homoplasmic mutant form, confirming the clinical suspicion of LHON and therefore Idebenone supplementation was initiated at 900 mg per day.The patient experienced progressive bilateral vision loss up to approximately 8 months due to an enlargement of the central scotoma at VF. During the early follow-up period (up to 8 months after the onset of the visual symptoms), a central GC-IPL thinning progressively extended following a characteristic pattern, as previously described in LHON, and progressive peripapillary RNFL thinning was most evident in the inferior and temporal sectors.1 After about 8 months from onset the patient experienced the nadir of VA with 0.02 OD and 0.04 OS associated with a large and dense ceco-central scotoma on VF (Figure 1). Ophthalmoscopy showed diffuse pallor of the optic nerves with a slight preservation of retinal nerve fibers (RNF) in the nasal sectors. Approximately one year after onset, the patient showed a slight, non-subjective improvement in VA and HVF (Figure 1) which then stabilized in the following months. After 3 years of Idebenone therapy at the dosage of 900 mg/die, it was decided to discontinue therapy, however the patient was followed-up over time with periodic ophthalmic examinations.

Figure 1. Visual field (VF) and visual acuity (VA) examinations in the right (OD) and left (OS) eyes at different timepoints. The impairment of OD was first detected and followed one month later by OS. The figure indicates the start of idebenone therapy, the different time points of VA and VF recovery, and the end of the recovery period. The 30° and 24° visual fields show the progressive enlargement of the central scotoma followed by its gradual reduction, whereas the 10° visual fields illustrate the fenestration of the central scotoma in the later stages.
Without any modification to his lifestyle habits, including diet and physical activity, the patient displayed a progressive improvement in visual function and at the visit conducted approximately four years after onset (one year after withdrawal of therapy) he presented with VA of 0.125 OD and 0.16 OS associated with a reduction of the ceco-central scotoma on HVF (Figure 1). Visual improvement continued for about 2.5 years. Five years after onset, VA reached 0.32 in OD and 0.4 in OS. At 5.8 years, VA improved to 0.63 OD and 0.63 OS, and by 6.8 years from onset it reached 0.8 OD and 0.8 OS. (Figure1) Concomitant with the improvement in VA, the 30° visual field showed relative stability, without evidence of further contraction of the scotoma.
The patient, who had consistently demonstrated a bilateral color vision deficit (as assessed by HRR testing 0/6 plates), did not show any improvement in color throughout follow-up.
At the most recent follow-up, approximately 7 years after onset, the patient maintained a stable VA of about 0.8 in both eyes. VF testing showed a persistent central scotoma, more extensive OS, with a small fenestration within the central 10° of the VF, which was also more evident OS. (Figure 1).
OCT analysis at 10 years from onset showed a diffuse reduction of RNFL and GCL thickness, which was more pronounced OD for RNFL and OS for GCL. The structural OCT findings can be correlated with the functional results, which demonstrated a slightly better visual field in OD and better VA in OS (Figure 2).2,3
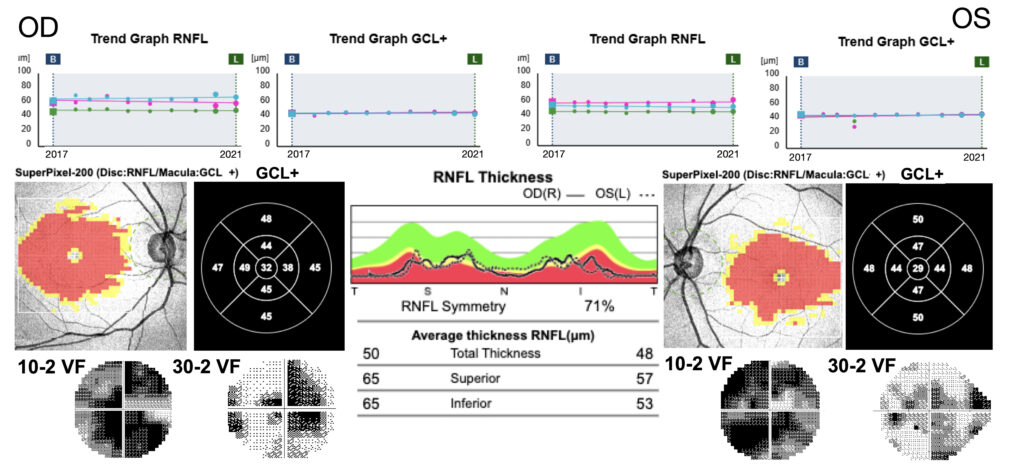
Figure 2. Retinal nerve fiber layer (RNFL) and ganglion cell layer (GCL+) thickness maps OD and OS at different timepoints (upper panel). The graphs demonstrate a stability of RNFL and GCL+ thickening through the follow-up visits from 3 yrs to 7 yrs from onset. In the middle panel are represented the RNFL and GCL+ thickness graphs which show the greater relative preservation of RNFL thickness in RE and GCL+ in LE in comparison with the 10° and 30° VF (bottom panel).
How can we explain the delayed functional recovery in the course of LHON?
The mechanisms underlying functional visual recovery, particularly when it occurs several years after disease onset in LHON, remain incompletely understood and challenging to fully elucidate.
Several potential mechanisms may be considered, including: (1) a delayed therapeutic effect of idebenone beyond its withdrawal, (2) neuroplasticity of cortical or visuospatial network remodeling, and (3) possible remyelination of surviving retinal ganglion cell axons. These mechanisms are not necessarily mutually exclusive and may act synergistically to support late functional recovery.
1) Idebenone therapy has been reported to achieve clinically meaningful stabilization of visual acuity in 46% of LHON patients over an average treatment duration of 25.6 months.4
Recently, a new ‘later is better’ paradigm has emerged, indicating that patients treated at the end of the dynamic phase of the disease (6 months from onset) exhibit a more favorable visual prognosis than those treated earlier. In this context, it can be hypothesized that under certain unknown conditions the pharmacological effects of idebenone require a prolonged period even beyond its withdrawal to exert clinically appreciable benefits as compared to the standard timing, which is after about two years administration as evidenced by the extended access program study and the LEROS trial.5-7
The idebenone therapeutic effect may take time as also evidenced in patients with another condition, dominant optic atrophy (DOA), with recovery of visual function detected at least after 7 months of treatment. Unlike LHON, DOA does not have a subacute phase which is now proposed to represent a refractory period to therapeutic interventions either with idebenone or gene therapy.8 Thus, considering that idebenone therapeutic effect needs a certain amount of time, we assume that residual viable fibers must be present, together with the potential for a late remyelination response. Ultimately, timing of idebenone therapeutic effect may be very variable on individual basis, with some cases significantly delayed.
Furthermore, a possible contributor to the patient’s favorable prognosis is the fact that idebenone’s efficacy depends on the flavoprotein NAD(P)H:quinone oxidoreductase (NQO1), which reduces idebenone to its active, electron-shuttling form.9 In this patient, the NQO1 enzyme genotype was WT/WT, exhibiting normal catalytic activity that likely contributed to the favorable outcome.
2) Some evidence supports also the hypothesis of cortical and visuospatial network neuroplasticity, indicating that brain reorganization can occur both following stroke and in cases of central visual function loss.10-12 Central vision loss or strokes both engage multiple neuronal mechanisms, including local retinotopic remapping, functional expansion of residual cortical regions, and strengthened connectivity and coordination between dorsal and frontal networks. All of these neural processes may underlie a potential recovery of visual function.
3) While antioxidant therapy with Idebenone may have partially preserved energy production in the surviving ganglion cells, a further event such as remyelination may have enabled the re-establishment of functional neuronal signaling. In LHON, surviving ganglion cells with impaired electrical activity due to energetic dysfunction undergo demyelination, which further diminishes the activation of these compromised axons. Within this vicious cycle, which renders the cells electrically silent, oligodendrocyte-mediated myelin remodeling could potentially restore the function of some ganglion cells, creating small fenestrations in the visual scotoma and thereby markedly improving the patient’s functional status without detectable structural changes.13
How can the differing recovery patterns of visual acuity and visual field be explained?
It has been demonstrated that smaller-caliber axons are particularly vulnerable in LHON, indicating that axon size plays a key role in resilience to metabolic stress.14,15 Based on this mechanism, it can be hypothesized that larger fibers are more prone to recover function through the restoration of metabolic activity, whereas smaller fibers preferentially degenerate and those surviving may recover firing of axonal potentials via remyelination processes. However, it remains difficult to understand and explain how some small fibers can persist beyond the acute phase of the disease. Visual field recovery appears to be primarily associated with the function of larger fibers, while improvements in visual acuity are linked to the restoration of smaller fibers within the papillomacular bundle, which mediates central vision. Recovery of function in LHON should be assessed using both visual acuity and visual field measurements, as they reflect two distinct, although partially overlapping, processes involving different anatomical structures.
It is, however, very difficult to explain the two-step recovery process observed in the patient at 1 year and 4 years after the onset of visual loss.
Conclusions
The present case illustrates the potential for a dual pattern of visual field and visual acuity recovery, characterized by delayed functional restoration of the small fibers in the papillomacular bundle.
ECM
References
- Balducci N, Savini G, Cascavilla ML, et al. Macular nerve fibre and ganglion cell layer changes in acute Leber’s hereditary optic neuropathy. Br J Ophthalmol. 2016 Sep;100(9):1232-7.
- Battista M, Coutinho CP, Berni A, et al. Sectorial Ganglion Cell Complex Thickness as Biomarker of Vision Outcome in Patients With Dominant Optic Atrophy. Invest Ophthalmol Vis Sci. 2024 Jan 2;65(1):24.
- Sergott RC, Carelli V, Newman NJ, et al. Predictors of Final Visual Outcome in Patients With Leber Hereditary Optic Neuropathy Treated With Lenadogene Nolparvovec Gene Therapy. Invest Ophthalmol Vis Sci. 2025 Jul 1;66(9):42.
- Catarino CB, von Livonius B, Priglinger C et al. Real-World Clinical Experience With Idebenone in the Treatment of Leber Hereditary Optic Neuropathy. J Neuroophthalmol. 2020 Dec;40(4):558-565.
- Carbonelli M, Amore G , Klopstock et al. Therapeutic intervention in Leber Hereditary Optic Neuropathy: later is better?. Presented at the 2025 ARVO Annual Meeting. Abstract IOVS June 2025, Vol.66, 3190.
- Newman NJ, Yu-Wai-Man P, Carelli V et al. Efficacy and safety of intravitreal gene therapy for leber hereditary optic neuropathy treated within 6 Months of disease onset. Ophthalmology. 2021:S0161-6420(20) 31187-8.
- Yu-Wai-Man P, Carelli V, Newman NJ, et al. Therapeutic benefit of idebenone in patients with Leber hereditary optic neuropathy: The LEROS nonrandomized controlled trial. Cell Rep Med. 2024 Mar 19;5(3):101437.
- Barboni P, Valentino ML, La Morgia C, et al. Idebenone treatment in patients with OPA1-mutant dominant optic atrophy. Brain. 2013 Feb;136(Pt 2):e231.
- Aleo SJ, Del Dotto V, Romagnoli M et al. Genetic variants affecting NQO1 protein levels impact the efficacy of idebenone treatment in Leber hereditary optic neuropathy. Cell Rep Med. 2024 Feb 20;5(2):101383.
- Payne BR, Lomber SG. Plasticity of the visual cortex after injury: what’s different about the young brain? Neuroscientist. 2002 Apr;8(2):174-85.
- Vasalauskaite A, Morgan JE, Sengpiel F. Plasticity in Adult Mouse Visual Cortex Following Optic Nerve Injury. Cereb Cortex. 2019 Apr 1;29(4):1767-1777.
- Cheung SH, Legge GE. Functional and cortical adaptations to central vision loss. Vis Neurosci. 2005 Mar-Apr;22(2):187-201.
- Chan JW, Sultan W, Karanjia R et al. Altering neuronal circuitry with 4-aminopyridine for visual improvement in Leber’s hereditary optic neuropathy (LHON). Mitochondrion. 2022 Jan;62:181-186.
- Sadun, A.A., Win, P.H., Ross‑Cisneros, F.N., Walker, S.O., Carelli, V. Leber’s hereditary optic neuropathy differentially affects smaller axons in the optic nerve. Transactions of the American Ophthalmological Society, 2000; 98: 223–232.
- Yu‑Wai‑Man, P., et al. A neurodegenerative perspective on mitochondrial optic neuropathies. Acta Neuropathologica, 2016.Dec;132(6):789-806.
