Luigi Brotto, Andrea Lembo, Paolo Nucci, University Eye Clinic San Giuseppe Hospital, University of Milan, Italy
A 3-year-old girl, first presented in June 2016 when her parents noticed marked photophobia and a tendency to hold objects very close to her face. At presentation, visual acuity measured with Lea symbols was 0.1 in the right eye and 0.4 in the left eye. Cover testing revealed intermittent exotropia of the right eye. Ocular motility examination showed no evident limitations of eye movements and stereopsis was absent. Anterior segment examination was unremarkable, and cycloplegic refraction revealed mild hyperopia, measured +2.25 diopters sphere with +0.75 diopters cylinder at 120° in the right eye (OD) and +2.50 diopters sphere in the left eye (OS). Fundus examination revealed elevated optic disc margins, more pronounced in the right eye with slight nasal sparing. Given the asymmetric optic disc swelling, an admission to neurology was advised.
During hospitalization, the neurological examination was within normal limits. Therefore, visual evoked potentials (VEP) and neuroimaging were indicated. VEP showed altered retino-cortical transmission and altered cortical activation. Ocular Ultrasound revealed mildly increased fluid in the optic nerve sheaths without evidence of optic nerve drusen. Brain and orbital MRIs with and without contrast showed that the optic nerves were slightly tortuous with mild distension of their cerebrospinal fluid sheaths, without abnormal contrast enhancement. Cerebral venous MR angiography and spinal MRI, with and without contrast, were unremarkable.
Inflammatory markers were negative, and autoimmune screening was unremarkable. Blood and CSF studies showed no evidence of systemic or central nervous system infection. CSF opening pressure was 20 cmH₂O, and the cerebrospinal fluid was normal.
The diagnosis of pseudotumor cerebri was suspected, and the patient was therefore discharged on Acetazolamide 125 mg twice daily.
Four months later, due to the severity of bilateral visual impairment (OD>OS) and persistent optic disc swelling with mild temporal pallor, the patient underwent an ex-juvantibus trial of high-dose corticosteroids, with intravenous methylprednisolone pulses at 30 mg/kg/day for 5 days followed by an oral prednisone taper over 2 weeks. Given the lack of response to acetazolamide, pseudotumor cerebri was considered unlikely, and this therapy was gradually discontinued. The differential diagnosis remained oriented towards hereditary optic neuropathies, including DOA and LHON.
Neuro-ophthalmologiy was consulted in December 2016, 6 months from onset; visual acuity was 0.06 in the right eye and 1.25 in the left eye at 1 meter. Ocular motility examination revealed an exotropia of the right eye of approximately 8 prism diopters for both distance and near, with absent stereopsis and a head tilt toward the left shoulder (figure1A). Pupils were equal and reactive to light, with no relative afferent pupillary defect (RAPD). Color vision could not be tested.
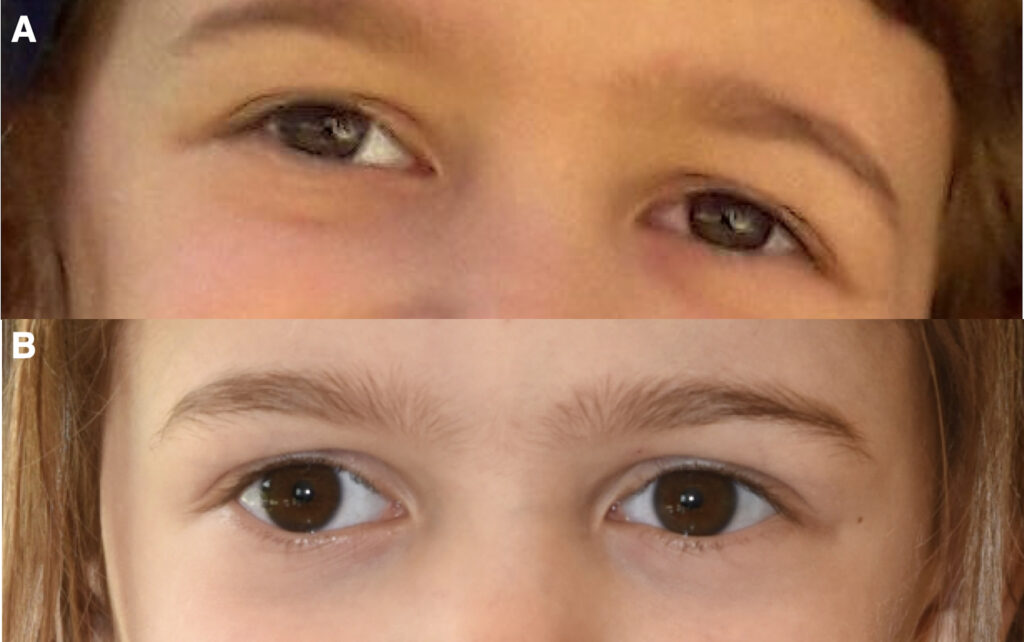
Figure 1 Photographs demonstrate right eye exotropia and compensatory head tilt toward the left shoulder at six months after the onset of visual loss (A) and the disappearance of the head tilt at the final follow-up examination (B).
Fundus examination revealed a mildly elevated optic disc consistent with pseudoedema, associated with subtle microangiopathic changes and a normal macula (figure 2).
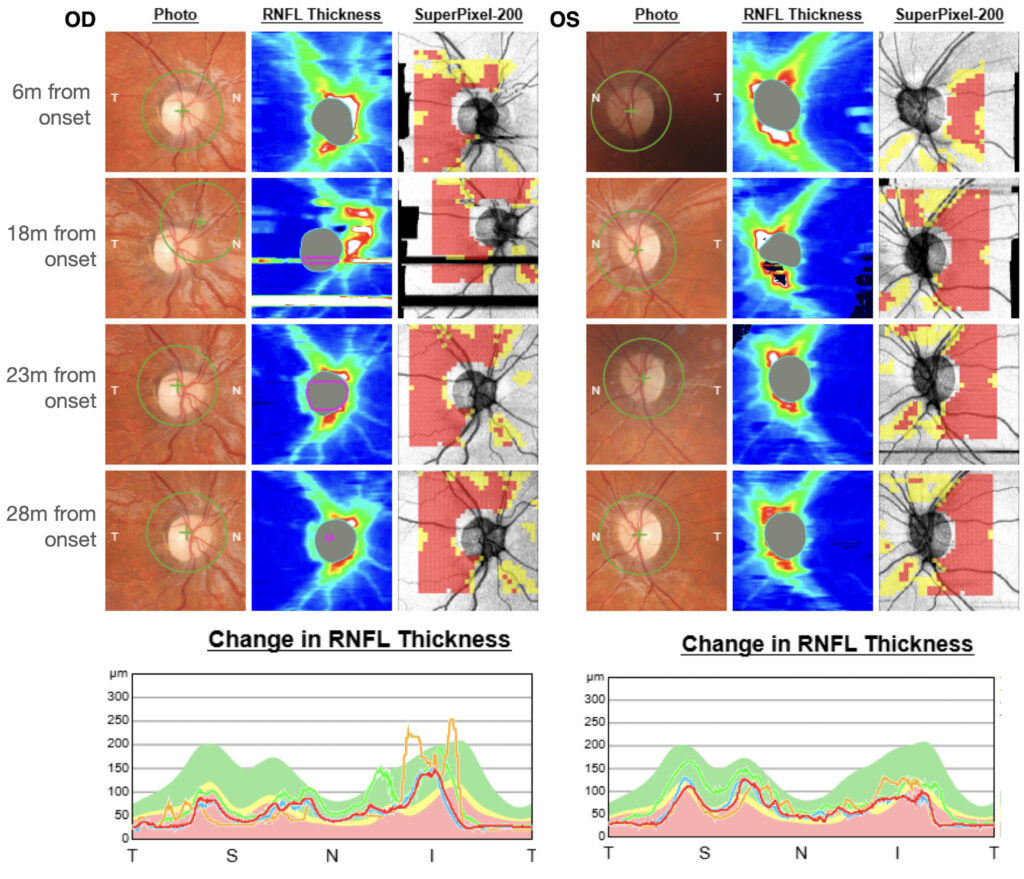
Figure 2: At baseline (6 months from onset), the average RNFL thickness was lower in the right eye compared to the left. It is possible that, at baseline, the right eye was already in the chronic phase, with a rapid progression of damage, which may have led to the development of exotropia in the right eye.
Genetic testing subsequently confirmed a homoplasmic m.3460G>A LHON mutation, establishing the diagnosis. The patient was initiated on idebenone therapy at a dose of 75 mg 6 times daily (half cps). Early treatment was complicated by gastrointestinal intolerance and transient elevation of liver enzymes, necessitating a dose adjustment to 75 mg three times daily.
One year and 7 months after symptom onset the patient underwent a comprehensive strabismus evaluation, which led to the suggestion of occlusion therapy of the left eye for six hours per day. Initially, the patient performed the patching with some difficulty but adhered to the regimen regularly. Over the subsequent years, occlusion was continued at a reduced daily duration of approximately two hours per day, performed intermittently as tolerated. Despite adherence, visual improvement remained limited. The occlusion therapy was ultimately discontinued approximately 3 years and 8 months after symptom onset, as continued patching provided no additional functional benefit.
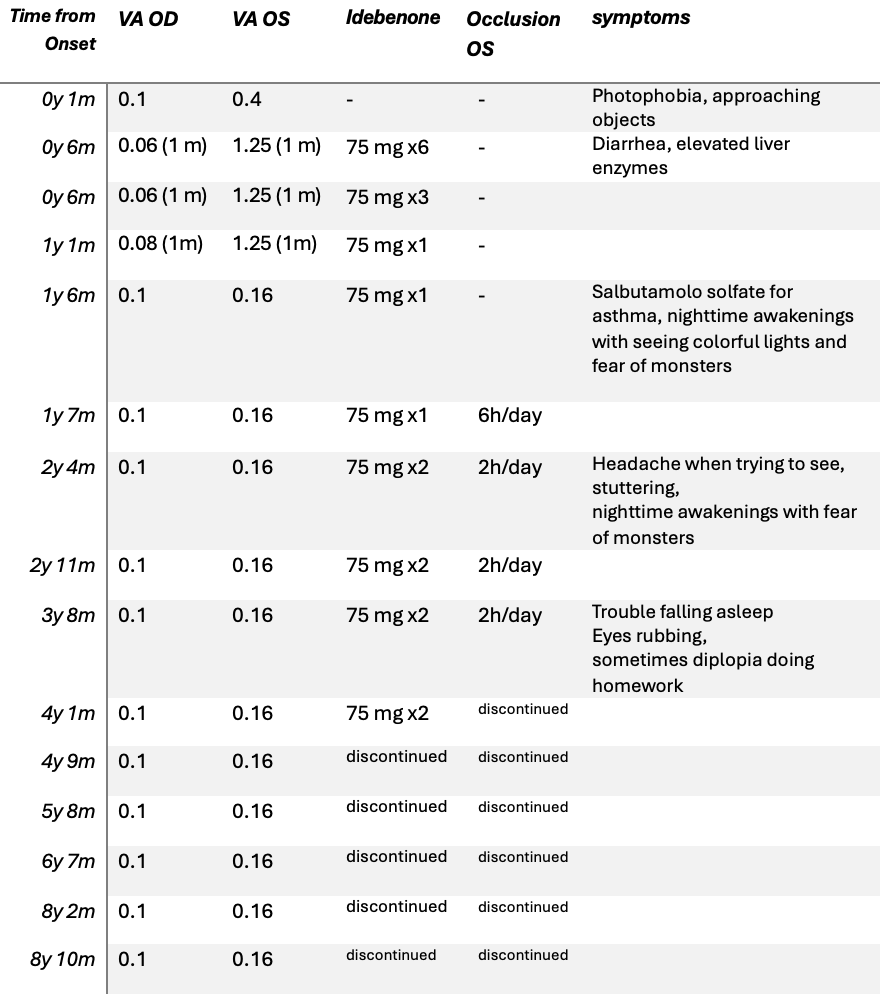
At 4 years and 6 months after presentation, the patient remained off idebenone. Visual acuity stabilized at low but relatively steady levels, 0.1 in the right eye and 0.16 in the left. She continued to experience slow reading due to scotomas, poor contrast sensitivity, and marked photophobia. The girl was hyperkinetic and highly aware of her visual condition. Throughout the clinical course, the young patient has reported multiple symptoms associated with her visual condition, including difficulty falling asleep (”until the little spots fall asleep”); nighttime awakenings with perception of a central white spot (the same spot appears black during the day) and seeing colorful lights and flashes. At school, she has trouble reading and wants to complete tasks quickly, which leads to frustration when she is unable to do so. With a keen interest in drawing, she expressed her perception of vision through her artwork (Figure 3).
Conclusion
At her most recent examination in April 2025, now aged 11, her condition appeared stable.
Visual acuity was unchanged at 0.1 in the right eye and 0.16 in the left. She still displayed mild variable exotropia with suppression of the right eye, and her optic discs showed diffuse pallor without excavation (Figure 4).

Figure 3: The figure presents two representative drawings by the patient, illustrating her subjective visual perception. In the lower drawing, she highlights the difference in visual experience between the two eyes.
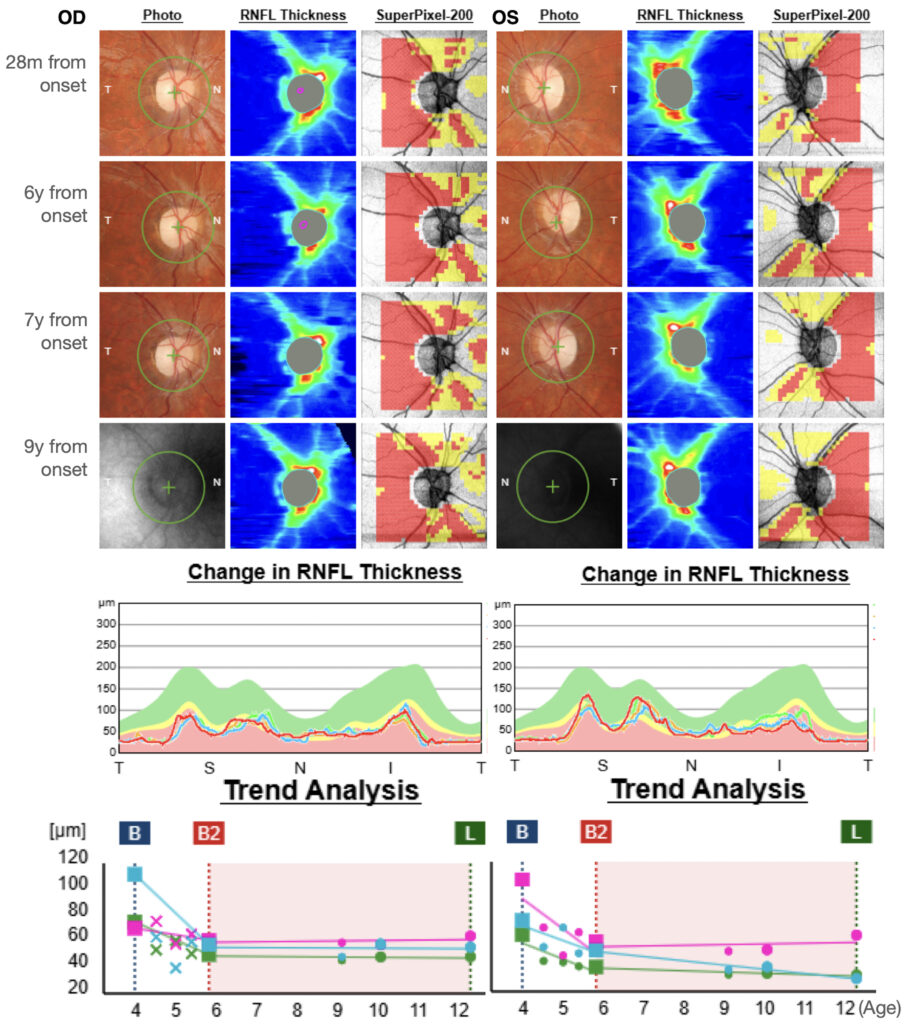
Figure 4: Optic nerve head at fundus examination and OCT of the optic nerve. Progression of retinal nerve fiber layer (RNFL) in the right (RE) and left eye (LE) at different timepoints of the follow-up. At 28 months from onset, a substantial stability of RNFL thickness was observed, with a slight progressive thinning of the inferior RNFL sector in the left eye. At the last follow-up visit, RNFL in OS remained thicker than OD (presence of fixation artifacts).
Discussion
Infrequently, LHON manifests in childhood, with onset before the age of 12 years. This pediatric form differs from the classical presentation in several ways: visual loss may follow a subacute, insidious, subclinical, or even unilateral course1.
As a subacute bilateral case, the onset of visual loss was clearly recognizable and evolved over a relatively short period, resembling the classical adult presentation of LHON. In these instances, children often reported a noticeable decrease in vision themselves, or the problem will be observed by parents or relatives.
Conversely, in insidious bilateral cases, the onset may be poorly defined. The decline in visual function frequently goes unnoticed by the family and is discovered incidentally during routine eye examinations, school screenings, or sports activities.
Diagnostic delays are frequent, as the condition is often initially mistaken for optic neuritis, compressive optic neuropathy, amblyopia, or functional visual loss. Despite these challenges, the visual prognosis in children under 9 years of age is generally better than in adults 2,3.
When suspect other disease form amblyopia?
In children, distinguishing functional amblyopia from optic nerve or retinal disease can be challenging, especially when the clinical presentation is subtle or asymmetric.
Functional amblyopia is, by definition, a reduction of visual acuity in the absence of detectable structural abnormalities in the eye or visual pathways. The fundus appears normal, OCT shows preserved RNFL and GCL profiles, and electrophysiological testing (VEP, ERG) reveals normal or near-normal responses.
Conversely, organic amblyopia refers to a reduction in visual acuity caused by structural damage to the eye, retina, or optic nerve, rather than by a functional cortical processing deficit. Optic neuropathies (hereditary, inflammatory, compressive, or ischemic) are characterized by objective structural or functional alterations, such as optic disc pallor, swelling, RNFL/GCL thinning, or abnormal visual evoked potentials.
The traditional distinction between functional and organic amblyopia has recently been reconsidered. Evidence now shows that even in so-called irreversible organic forms, some functional improvement is possible. Conversely, purely functional amblyopia has been associated with subtle structural changes in the lateral geniculate nucleus and primary visual cortex, suggesting that both forms involve a combination of functional and anatomical alterations.
Why does she have a head tilt?
In cases of sensory exotropia, an abnormal head posture is not consistently present. However, it may occasionally develop as a compensatory mechanism. When one eye has reduced visual function, the patient may adopt a slight head tilt or turn toward the better-seeing eye to optimize monocular vision and improve apparent ocular alignment. In this context, the abnormal head posture serves primarily as a functional adaptation rather than reflecting a primary oculomotor deficit.
In cases of sensory exotropia, a mild head tilt may develop as a compensatory mechanism to optimize fixation with the better-seeing eye. As visual function stabilizes and suppression of the poorer eye becomes consistent, this abnormal head posture may gradually disappear, reflecting functional adaptation rather than improvement of extraocular motility.
What is the effect of idebenone in children with LHON?
The role of idebenone in pediatric LHON remains an area of clinical uncertainty. Idebenone, a short-chain benzoquinone that acts as an electron carrier bypassing complex I deficiency, has been approved by the European Medicines Agency (EMA) for the treatment of visual impairment in adolescents and adults with LHON, but its formal indication does not extend below the age of 12, largely due to the absence of randomized controlled trial data in younger children. The seminal RHODOS trial enrolled only patients aged 14 years and above4.
Nevertheless, pediatric experience is growing through case reports and small case series, suggesting that idebenone may be both safe and potentially effective when administered early after disease onset. In children, the pathophysiological rationale for treatment may be even stronger than in adults: the optic nerve is less atrophic in the acute phase, mitochondrial plasticity is higher, and the visual system retains greater capacity for recovery.
In the natural history of pediatric LHON, patients aged 9 to 12 years tend to exhibit a disease course comparable to that observed in adults, whereas those younger than 9 years generally demonstrate a more favorable visual prognosis. Consequently, the necessity and efficacy of idebenone treatment in children under 9 years of age remain uncertain1.
From a safety perspective, idebenone has generally been well tolerated; most patients do not experience significant adverse effects. The most frequently reported side effects are mild gastrointestinal disturbances, such as abdominal discomfort, diarrhoea, or nausea. These events are typically transient, self-limiting, and rarely require discontinuation of therapy.
Is strabismus a negative prognostic factor in these cases?
Strabismus is a frequent and generally nonspecific finding in children, yet it appears consistently in almost all reported unilateral cases of childhood LHON, where it is usually associated with profound visual loss. In affected families, the presence of strabismus may therefore signal an underlying optic neuropathy. Interestingly, the fellow eye can remain unaffected until adolescence or be involved only subclinically, a pattern rarely encountered in adult-onset LHON. This unusual asymmetric onset in infancy may be linked to subtle anatomical differences between the eyes, such as variations in optic nerve head morphology or axonal number1. The development of strabismus in these cases may be age-dependent: sensory deprivation in one eye during critical periods of visual development can disrupt normal binocular function, particularly in unilateral or asynchronously affected eyes, leading to compensatory ocular misalignment.
Is it appropriate to treat functional amblyopia in LHON strabismus?
Treating functional amblyopia in patients with LHON-related strabismus presents unique therapeutic challenges. Unlike typical strabismus-related amblyopia in otherwise healthy eyes, the visual system in these patients has pre-existing structural or functional deficits, which may limit cortical plasticity and reduce the efficacy of conventional occlusion therapy. Patching therapy may still confer some benefit in sensory amblyopia, although the improvement is often limited and variable. The degree of improvement depends on the residual visual function, the duration of deprivation, and the timing of intervention.
The rationale behind patching in sensory amblyopia is primarily to maximize the use of the weaker eye, thereby stimulating residual cortical pathways. However, because suppression in these cases often arises from sensory deprivation rather than abnormal binocular interaction, treatment protocols need to be carefully tailored.
Intermittent occlusion schedules, shorter daily durations, or partial patching may be more tolerable than prolonged full-time patching, which is often very difficult to tolerate due to the poor residual visual function. However, a potential recovery after prolonged patching cannot be excluded, considering the degree of neuroplasticity at this age, even though the residual RNFL thickness in OD was lower compared to the fellow eye.
Timing and age-dependence are crucial considerations. Neuroplasticity declines with age, so earlier intervention increases the likelihood of functional gains. Even limited improvements, however, can significantly enhance quality of life by reducing eccentric viewing, improving fixation stability, and facilitating activities of daily living.
Conclusion
This is the story of a remarkable little girl with an eye condition that changed her perception of the world around her, yet she faced life with an indomitable spirit and the support of a family that surrounded her with love. They guided her through challenges with the infinite care of dedicated parents—stepping back when needed and offering support when necessary—watching her grow with the eyes that only parents have for their child.
This spirited girl, with her sensitivity and boundless energy, captivated all the doctors and staff.She gifted us not only her “visionary” drawings but also a lesson in how to truly see life.
ECM
Bibliography
- Barboni P, La Morgia C, Cascavilla ML, Hong EH, Battista M, Majander A, Caporali L, Starace V, Amore G, Renzo AD, Carbonelli M, Nucci P, Jurkute N, Chen BS, Panebianco R, De Negri AM, Sadun F, Parisi V, Bandello F, Sadun AA, Carelli V, Yu-Wai-Man P. Childhood-Onset Leber Hereditary Optic Neuropathy – Clinical and Prognostic Insights. Am J Ophthalmol. 2023 May;249:99–107. doi: 10.1016/j.ajo.2022.12.014. Epub 2022 Dec 18. PMID: 36543315.
- Barboni P, Carbonelli M, Savini G, Ramos Cdo V, Carta A, Berezovsky A, Salomao SR, Carelli V, Sadun AA. Natural history of Leber’s hereditary optic neuropathy: longitudinal analysis of the retinal nerve fiber layer by optical coherence tomography. Ophthalmology. 2010 Mar;117(3):623–627. doi: 10.1016/j.ophtha.2009.07.026. Epub 2010 Jan 19. PMID: 20031228.
- Majander A, Bowman R, Poulton J, Antcliff RJ, Reddy MA, Michaelides M, Webster AR, Chinnery PF, Votruba M, Moore AT, Yu-Wai-Man P. Childhood-onset Leber hereditary optic neuropathy. Br J Ophthalmol. 2017 Nov;101(11):1505–1509. doi:10.1136/bjophthalmol-2016-310072. Epub 2017 Mar 17. PMID: 28314831.
- Klopstock T, Yu-Wai-Man P, Dimitriadis K, Rouleau J, Heck S, Bailie M, Atawan A, Chattopadhyay S, Schubert M, Garip A, Kernt M, Petraki D, Rummey C, Leinonen M, Metz G, Griffiths PG, Meier T, Chinnery PF. A randomized placebo-controlled trial of idebenone in Leber’s hereditary optic neuropathy.Brain. 2011 Sep;134(Pt 9):2677–2686. doi:10.1093/brain/awr170. Epub 2011 Jul 25. PMID: 21788663; PMCID: PMC3170530.
