Piero Barboni, Scientific Institute San Raffaele Hospital, Milan, Italy and Studio Oculistico d’Azeglio, Bologna, Italy
Valerio Carelli, Department Biomedical and Neuromotor Sciences, University of Bologna, Bologna, Italy and IRCCS, Istituto delle Scienze Neurologiche, Programma di Neurogenetica, Bologna, Italy
A 60-year-old male patient came to the clinic to revaluating his optic neuropathy that had never been clearly defined. At the age of 19 (on March 1983), he experienced vision loss in one eye, which was followed by involvement of the fellow eye one month later. He was the youngest of eight siblings, with four brothers. Notably, one of his brothers had a similar episode a decade earlier, also occurring at the age of 20.
Following his brother’s experience with surgery for opto-chiasmatic arachnoiditis in Geneva in 1972, he underwent the same procedure after an intravenous steroid therapy failed to promote any improvement.
The surgery, consisting of ‘inspection of the optic chiasm and removal of the myelin sheaths’, was performed in August 1983. Similar to his brother, he the experienced visual recovery, which he reported to have occurred in April 1984.
Clinical case description
On examination, visual acuity was 0.6 in the right eye (OD) and 1.0 in the left eye (OS). Color vision was 0/6 in both eyes on HRR plates. IOP was 14mmHg in OU. Pupils were normally reactive. Visual field testing revealed a small ceco-central scotoma in both eyes, more prominent in OD (Figure 1A). Fundus examination showed an enlarged optic disc with temporal pallor (Figure 1B). OCT imaging demonstrated temporal RNFL thinning, corresponding to a diffuse reduction of the macular GCL (Figure 1C).
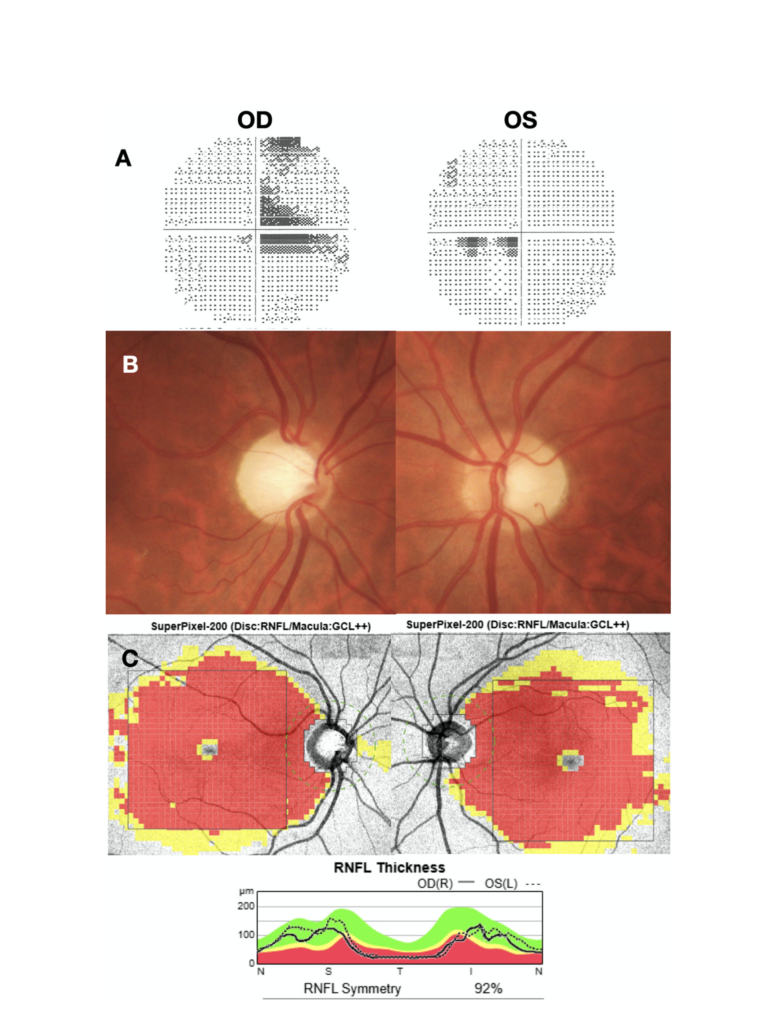
Figure 1.
A. Visual field analysis showing a small ceco-central scotoma in both eyes, more prominent in the right eye (OD).
B. Fundus examination of the optic nerve head showing increased and asymmetric cupping, greater in the right eye (OD) than in the left eye (OS), associated with temporal pallor.
C. Optic nerve OCT demonstrating temporal thinning of the retinal nerve fiber layer (RNFL) and diffuse thinning of the macular ganglion cell layer (GCL) in both eyes, more extended in OD.
The patient’s clinical history immediately took me back to the years when I was studying ophthalmology, specifically to 1988, when, following Doug Wallace’s article, I started studying a disease called Leber’s hereditary optic neuropathy (LHON).1 At that time, I had begun working in molecular biology in the laboratory my father had established in the hospital to perform the first PCR experiments. We had designed primers to diagnose LHON, specifically targeting the m.11778G>A mutation. However, we had no patients with LHON available. When a friend of mine informed me that the neurology department was following a large affected family, I immediately asked whether we could obtain a blood sample for testing.
After making the diagnosis in our first LHON family, my friend Valerio and I began to broaden our focus to mitochondrial diseases.2 While he pursued a distinguished academic career in mitochondrial medicine, I explored the ophthalmology clinic in search of patients who might have mitochondrial disorders. In reviewing the extensive departmental records, I encountered a rare condition called opto-chiasmatic arachnoiditis, which displayed ophthalmologic features strikingly reminiscent of those seen in LHON, and most of them underwent the same surgical procedure.
While reviewing the medical records and investigating patients with arachnoiditis, I came across several families affected by this condition, with multiple cases recurring within the same family.
I reached out to all the families and we screened for the LHON-associated mutations known at the time. When the genetic results came back, we were stunned: every single family turned out to be affected by LHON!
Since that time, we have continued our work on these diseases, caring for around 200 families with LHON, as well as a similar number affected by other mitochondrial or nuclear DNA disorders—such as dominant optic atrophy or the recessive form of LHON, to name just a few.3-5
Continuing with the case description, mitochondrial DNA testing was requested based on the earlier suspicion of LHON. The analysis confirmed the combination of two heteroplasmic variants m.14258G>A; p.Pro139Leu and m.14582A>G; p.Val31Ala of MT-ND6 in a mitochondrial haplogroup K1a background. This rare combination of variants was described by me and Valerio in 2018 with Leonardo Caporali, as all these patients belong to a single large cluster of maternally inherited LHON originating from southern Italy.6
In conclusion, we describe two brothers diagnosed with arachnoiditis in the early 1980s who experienced visual improvement following neurosurgical intervention. These cases add to several others reported during that period in which patients underwent the same surgical procedure and showed similar visual recovery. The subsequent diagnosis of LHON raises a key question: why would a surgical intervention lead to visual improvement in patients with LHON?
How many cases have truly been mistaken for LHON?
Looking back at idiopathic opto-chiasmatic arachnoiditis, this disease marked the start of our critical reassessment of a diagnosis that might never have been entirely correct, encompassing a spectrum of clinical forms, as recently highlighted.7 The clinical picture of acute, painless bilateral vision loss associated with presumed optic disc edema, in the absence of other symptoms, could at that time be classified as idiopathic opto-chiasmatic arachnoiditis. Several authors had already included Leber’s hereditary optic neuropathy in the differential diagnosis due to the overlap in clinical presentation especially when pseudoedema and microangiopathy were present.7,8
Could a surgical intervention improve the prognosis of LHON?
The diagnosis of arachnoiditis was made based on the surgical finding of arachnoid thickening. The surgical treatment consisted of releasing adhesions and draining excessive fluid. However, the benefits in the case of arachnoiditis were difficult to interpret, as one would expect the adhesions to reform over time.9-10
In the case of LHON, one could speculate that the surgical procedure may reduce compression and axoplasmic blockage. The procedure would therefore act on the mechanical component. Moreover, this overlap brings renewed attention to the recent hypothesis of an inflammatory component in LHON.
In this latter scenario, a histopathological rationale would be necessary to support such an approach. Ultimately, revisiting anatomical specimens from previous surgeries could offer valuable insights into the histopathology of LHON.
ECM
Principal Figures and Contributors in the 1988 Narrative
Dr. Piero Barboni, resident in ophthalmology, University of Bologna
Dr. Valerio Carelli, resident in neurology, University of Bologna
Prof. Francesco Barboni; head of Laboratorio analysis Malpighi Hospital, Bologna
Dr.ssa Wilma Mantovani, biologist, Malpighi Hospital, Bologna
Dr.ssa Giulia Pierangeli, neurologist Clinica Neurologica, University of Bologna
Prof Pasquale Montagna, neurologist Clinica Neurologica, University of Bologna
Dr. Pietro Cortelli, neurologist Clinica Neurologica, University of Bologna
Prof Gaist, Neurosurgeon Ospedale Bellaria, University of Bologna
Prof Bernei, Neurosurgeon, Geneva
References
- Wallace DC, Singh G, Lott MT, Hodge JA, Schurr TG, Lezza AM, Elsas LJ 2nd, Nikoskelainen EK. Mitochondrial DNA mutation associated with Leber’s hereditary optic neuropathy. Science. 1988 Dec 9;242(4884):1427-30.
- Cortelli P, Montagna P, Avoni P, Sangiorgi S, Bresolin N, Moggio M, Zaniol P, Mantovani V, Barboni P, Barbiroli B, et al. Leber’s hereditary optic neuropathy: genetic, biochemical, and phosphorus magnetic resonance spectroscopy study in an Italian family. Neurology. 1991 Aug;41(8):1211-5.
- Puddu P, Barboni P, Mantovani V, Montagna P, Cerullo A, Bragliani M, Molinotti C, Caramazza R. Retinitis pigmentosa, ataxia, and mental retardation associated with mitochondrial DNA mutation in an Italian family. Br J Ophthalmol. 1993 Feb;77(2):84-8.
- Fiorini C, Ormanbekova D, Palombo F, Carbonelli M, Amore G, Romagnoli M, d’Agati P, Valentino ML, Barboni P, Cascavilla ML, De Negri A, Sadun F, Carta A, Testa F, Petruzzella V, Guerriero S, Bianchi Marzoli S, Carelli V, La Morgia C, Caporali L. The Italian reappraisal of the most frequent genetic defects in hereditary optic neuropathies and the global top 10. Brain. 2023 Sep 1;146(9):e67-e70.
- Borrelli E, Bandello F, Boon CJF, Carelli V, Lenaers G, Reibaldi M, Sadda SR, Sadun AA, Sarraf D, Yu-Wai-Man P, Barboni P. Mitochondrial retinopathies and optic neuropathies: The impact of retinal imaging on modern understanding of pathogenesis, diagnosis, and management. Prog Retin Eye Res. 2024 Jul;101:101264.
- Caporali L, Iommarini L, La Morgia C, et al. Peculiar combinations of individually non-pathogenic missense mitochondrial DNA variants cause low penetrance Leber’s hereditary optic neuropathy. PLoS Genet. 2018 Feb 14;14(2):e1007210
- Lessell S, E Grzybowski A. Idiopathic opticochiasmatic arachnoiditis. J Neuroophthalmol. 2014 Sep;34(3):251-4.
- de Gottrau P, Büchi ER, Daicker B. Distended optic nerve sheaths in Leber’s hereditary optic neuropathy. J Clin Neuroophthalmol. 1992 Jun;12:89-93.
- Cogan DG. Neurology of the Visual System. Springfield, Illinois: Charles C. Thomas, 1966:217–218, 221.
- Walsh FB, Hoyt WF. Clinical Neuro-Ophthalmology. Baltimore MD: Williams and Wilkins; 1969;3:2100–2103.
- Lowes MM, Ehlers N, Malmros R. Recurrent chiasmal arachnoiditis successfully treated by operations. Acta Ophthalmol. 1974;52:885–892.
